Abstract
- NIMS and Hokkaido University jointly discovered that proton transfer in electrochemical reactions is governed by the quantum tunneling effect (QTE) under the specific conditions. In addition, they made a first ever observation of the transition between the quantum and classical regimes in electrochemical proton transfer by controlling potential. These results may advance basic research leading to the development of highly efficient electrochemical energy conversion systems based on quantum mechanics.
- Many of the state-of-the-art electronic devices and technologies that have realized present modern lives were established based on the fundamental principles of quantum mechanics. Quantum effects in electrochemical reactions in fuel cells and energy devices are, however, not well understood due to the complex movement of electrons and protons driven by electrochemical reaction processes on the surfaces of electrodes. As the result, application of quantum effects in electrochemical energy conversion is not as successful as the fields of electronics and spintronics, which surface and interfacial phenomena are equally critical in all of these fields. Assuming that electrochemical reactions are closely associated with quantum effects, it may be feasible to design highly efficient energy conversion mechanisms based on these effects: including QTE, and devices that take advantage of such mechanisms.
- In this study, the NIMS-led research team focused on oxygen reduction reaction (ORR) mechanisms—the key reaction in fuel cells—using deuterium, an isotope of hydrogen having a different mass. As the result, the team confirmed proton tunneling through activation barriers within a small overpotential range. Furthermore, the team found that an increase in overpotential leads to electrochemical reaction pathways to change to proton transfer based on the semiclassical theory. Thus, this research team discovered the novel physical processes: the transition between the quantum and classical regimes in electrochemical reactions.
(See Figure)Schematic diagram for two possible paths of the proton-transfer reaction
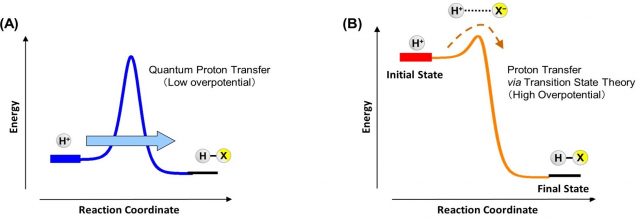
Figure: (A) Quantum tunneling of protons. (B) Protons passing through the transition state
(A) proton tunneling through the barrier (quantum). (B) proton transfer via the transition state (classical); In the electrochemical system the relative contribution of the two mechanisms can be tuned by the applied potential.
- This research shows the involvement of QTE in proton transfer during the basic energy conversion processes. This discovery may facilitate investigations of microscopic mechanisms of electrochemical reactions which are not understood in detail. It may also stimulate the development of highly efficient electrochemical energy conversion technology with a working principle based on quantum mechanics, capable of operating beyond the classical regime.
- This research project was conducted by a research team led by Ken Sakaushi (Senior Researcher, Center for Green Research on Energy and Environmental Materials [C4GR], NIMS), Andrey Lyalin (Special Researcher, C4GR, NIMS) and Tetsuya Taketsugu (Professor, Institute for Chemical Reaction Design and Discovery, Creative Research Institution, Hokkaido University). This project was supported by the JSPS Grant-in-Aid for Young Scientists (B) (Project No. 17K14546), the JSPS Grant-in-Aid for Scientific Research (C) (Project No. 15K05387) and the MEXT Program for Development of Environmental Technology using Nanotechnology.
This study was published in Physical Review Letters, a journal of the American Physical Society, on December 7, 2018.
Contacts
(Regarding this research)
Ken Sakaushi
Senior Researcher
Center for Green Research on Energy and Environmental Materials
National Institute for Materials Science
Tel: +81-29-860-4945
Email: SAKAUSHI.Ken=nims.go.jp
(Please change “=” to “@”)
URL: https://samurai.nims.go.jp/profiles/sakaushi_ken
Tetsuya Taketsugu
Professor
Quantum Chemistry Laboratory, Department of Chemistry, Faculty of Science, Hokkaido University;
Creative Research Institution; Institute for Chemical Reaction Design and Discovery
Tel: +81-11-706-3535
Email: take=sci.hokudai.ac.jp
(Please change “=” to “@”)
URL: https://wwwchem.sci.hokudai.ac.jp/~qc/
(For general inquiries)
Public Relations Office
National Institute for Materials Sciences
Tel: +81-29-859-2026 Fax: +81-29-859-2017
E-Mail: pressrelease=ml.nims.go.jp
(Please change “=” to “@”)
Public Relations Division
General Affairs and Planning Department, Hokkaido University
Kita 8, Nishi 5, Kita-ku, Sapporo, Hokkaido 060-0808, Japan
Tel: +81-11-706-2610 Fax: +81-11-706-2092
Email: kouhou@jimu.hokudai.ac.jp
“Quantum-to-Classical Transition of Proton Transfer in Potential-Induced Dioxygen Reduction”: Ken Sakaushi, Andrey Lyalin, Tetsuya Taketsugu, Kohei Uosakii: Physical Review Letters: DOI:10.1103/PhysRevLett.121.236001

