A team of scientists has succeeded in visualizing how carbon dioxide (CO2) behaves in an ionic liquid that selectively absorbs CO2. The finding is expected to help develop more efficient methods to capture CO2 in the atmosphere, one of the major factors causing global warming.
The CO2-absorbing soft crystal developed for this study (Photo: Shin-ichiro Noro).
Carbon dioxide (CO2) levels in the atmosphere — a major factor in global warming — continue to rise every year, creating grave concerns about the future of Earth. To halt global warming, our industrial society needs to emit much less CO2. One way of achieving this is to separate and collect CO2 before it is released into the atmosphere. While some such efforts are already underway, they have not been very efficient. There is thus an urgent need to develop technology that can separate and collect CO2 more efficiently, both to protect the environment but also to promote recycling of CO2 as a resource.
The use of ion liquids to effectively absorb CO2 has been the subject of intensive research. Yet more investigation of how CO2 absorbed in ionic liquids behaves is needed to improve the materials used in the CO2 separation and collection process. As ionic liquids are a fluid with no regular structure, it has been difficult to directly observe the state of CO2 absorbed in them.
In the present study, a team of scientists that included Professors Shin-ichiro Noro and Takayoshi Nakamura, both of Hokkaido University’s Graduate School of Environmental Science, focused on a soft crystal, a substance that possesses both the softness of a liquid and the regularity of a crystal. They synthesized a soft crystal containing components of an ionic liquid that absorbed CO2. As anticipated, the soft crystal maintained its regularity even after it absorbed CO2, making it possible to conduct X-ray diffraction analysis.
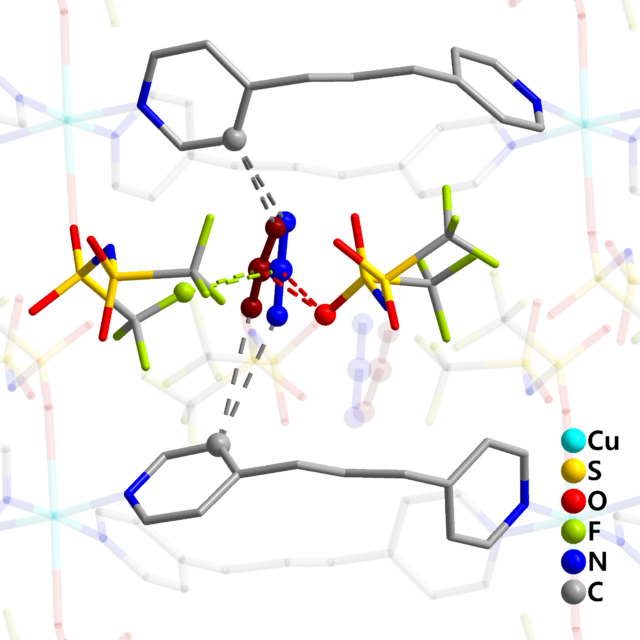
CO2 molecules (red and blue at the center), absorbed by the soft crystal, interact with both fluorine and oxygen atoms of a component of the ionic liquid, bis(trifluoromethylsulfonyl)imide
(Xin Zheng, et al, Communications Chemistry, October 27, 2020).
The analysis showed the absorbed CO2 interacts with both fluorine and oxygen atoms of the bis(trifluoromethylsulfonyl)imide anion, a component of the ionic liquid. Furthermore, the scientists’ theoretical analysis showed that dispersion and electrostatic interactions exist between CO2 and the framework, creating the force that binds CO2 to the anion.
The team’s findings are expected to be helpful in designing and developing ionic liquids capable of efficiently separating and collecting CO2, and will likely accelerate practical applications of such liquids, a pivotal step to alleviating the negative effects of global warming.
Shin-ichiro Noro focuses on the development of porous materials to contribute to environmental restoration and conservation, while Takayoshi Nakamura’s work is focused on the development of molecular devices for a wide variety of applications.
Researchers who contributed to this study:
(Back, from left) Takayoshi Nakamura, Shin-ichiro Noro, Jenny Pirillo,
(front, from left) Kiyonori Takahashi, Xin Zheng, and Yuh Hijikata (Photo: Shin-ichiro Noro).
Original article
Xin Zheng, et al. Understanding the interactions between the bis(trifluoromethylsulfonyl)imide anion and absorbed CO2 using X-ray diffraction analysis of a soft crystal surrogate. Communications Chemistry. October 27, 2020.
DOI: 10.1038/s42004-020-00390-1
Funding information
This study was supported financially by a Grant-in-Aid for Scientific Research (18K19864) and the Program for Leading Graduate Schools (Hokkaido University “Ambitious Leader’s Program”) from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT), Japan.
Contacts
Professor Shin-ichiro Noro
Graduate School of Environment Science, Hokkaido University
Email: noro[at]ees.hokudai.ac.jp
Sohail Keegan Pinto (International Public Relations Specialist)
Public Relations Division, Hokkaido University
Tel: +81-11-706-2185
Skype: hokudai.pr1
Email: en-press[at]general.hokudai.ac.jp