A new method for creating a highly useful chemical subunit eliminates the need for precious metals, potentially leading to the sustainable production of pharmaceuticals and electronics.
 Reaction vials being exposed to blue light from an LED. (Photo: Yusuke Masuda)
Reaction vials being exposed to blue light from an LED. (Photo: Yusuke Masuda)
Light-emitting diodes (LEDs) are commonly used in the electronic displays in our TVs, smartphones, and other devices; now, researchers at the Institute of Chemical Reaction Design and Discovery (ICReDD) at Hokkaido University have utilized blue LEDs to develop a more sustainable way to make key chemical subunits that have potential uses in pharmaceutical and photoelectronic development.
Researchers utilized blue LEDs in combination with a copper-based molecular catalyst to perform what is known as a cross-coupling reaction, in which two molecules are joined via a carbon-carbon bond. This is one of the most widely used types of reactions and is essential for creating most of the chemical products used today. The use of copper—a cheaper and more commonly available metal—as a catalyst for a cross-coupling reaction is a major breakthrough in sustainability, since this transformation typically relies on the use of precious metals such as palladium.
The new method is also advantageous because the copper metal in the molecular catalyst itself absorbs the blue light, rather than needing to add an additional light-absorbing compound in addition to the catalyst. This makes the synthesis not only cheaper and simpler to perform, but also easier to control, since there are fewer moving parts.
The blue light plays a key role in activating the copper-based catalyst. Theoretical calculations showed that this light exposure causes electrons to move from the metal copper atom to a connected subunit of the molecular catalyst. This excited state has separated electrical charges, making the catalyst much more reactive, and researchers were able to use it to carry out a cross-coupling reaction that creates an acyl group, which are useful for the synthesis of pharmaceuticals and photoelectronic materials.
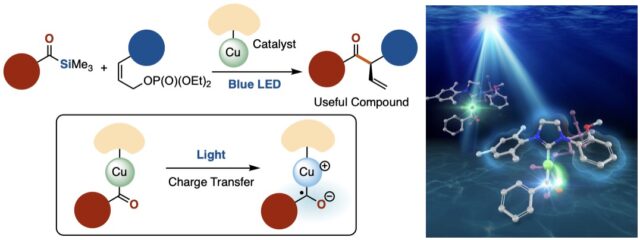 (Top Left) Schematic of cross-coupling reaction using a copper-based catalyst. (Bottom Left) Schematic showing the excitation of the copper-based catalyst by blue LED light and the resulting charge transfer. (Right) Artistic depiction of the catalyst absorbing light and being activated. (Yusuke Ueda, et al. Journal of the American Chemical Society. January 6, 2022).
(Top Left) Schematic of cross-coupling reaction using a copper-based catalyst. (Bottom Left) Schematic showing the excitation of the copper-based catalyst by blue LED light and the resulting charge transfer. (Right) Artistic depiction of the catalyst absorbing light and being activated. (Yusuke Ueda, et al. Journal of the American Chemical Society. January 6, 2022).
A key aspect of this method is that the formation of the acyl group occurs asymmetrically. This means one of the two possible mirror image versions of the product molecule is selectively produced, a feature highly desired for development of new medicines. Researchers tested their new method with multiple different starting materials, notably including a material derived from probenecid, a medicine for gout. The product obtained from this starting material has potential applications in the pharmaceutical industry. The high selectivity of this method and its compatibility with compounds used in medicine highlights its potential utility.
Implementation of this new method is expected to both provide cost savings and increase the sustainability of the production of a wide variety of chemical compounds with potential uses in medicine and electronics. Its utilization of commonly available materials makes it especially appealing.
“This synthetic method is a breakthrough because it combines two easily obtainable items, blue LED light and copper, to achieve a coupling reaction that did not exist before,” commented Assistant Professor Yusuke Masuda. “Technology that produces useful compounds from resources which are abundantly available on Earth is critical for the sustainable development of humanity. I expect this advance will become a milestone in the development of sustainable molecular synthetic methods.”
 Professor Masaya Sawamura (left), Professor Jun-ya Hasegawa (middle), and Assistant Professor Yusuke Masuda (right) of the research team at the Institute for Chemical Reaction Design and Discovery (ICReDD) and Hokkaido University. (Photos provided by ICReDD and Hokkaido University).
Professor Masaya Sawamura (left), Professor Jun-ya Hasegawa (middle), and Assistant Professor Yusuke Masuda (right) of the research team at the Institute for Chemical Reaction Design and Discovery (ICReDD) and Hokkaido University. (Photos provided by ICReDD and Hokkaido University).
Original Article:
Yusuke Ueda, et al. Photoinduced Copper-Catalyzed Asymmetric Acylation of Allylic Phosphates with Acylsilanes. J. Am. Chem. Soc. January 6, 2022. DOI: 10.1021/jacs.1c11526
Funding:
This work was supported by the following: JSPS KAKENHI Grant JP21H04680 in Grant-in-Aid for Scientific Research (A) (M.S., J.-y.H., K.U.), Grant JP18H03906 in Grant-in-Aid for Scientific Research (A) (M.S.), Grant JP21J11643 in Grant-in-Aid for JSPS Fellows (Y.U.), Grant JP21K14626 in Grant-in-Aid for Early-Career Scientists (Y.M.), Grant JP20H04793 in Grant-in-Aid for Scientific Research on Innovative Areas, “Hybrid Catalysis” (T.I.), and Grant JP19H02737 in Grant-in-Aid for Scientific Research (B) (K.U.).
Contacts:
Professor Masaya Sawamura
Institute for Chemical Reaction Design and Discovery; Department of Chemistry, Faculty of Science
Hokkaido University
E-mail: sawamura[at]sci.hokudai.ac.jp
Professor Jun-ya Hasegawa
Institute for Chemical Reaction Design and Discovery; Institute for Catalysis
Hokkaido University
E-mail: hasegawa[at]cat.hokudai.ac.jp
Collin Stecker (Public Relations and Outreach)
Institute for Chemical Reaction Design and Discovery
Hokkaido University
Tel: +81-11-706-9646
E-mail: public_relations[at]icredd.hokudai.ac.jp

