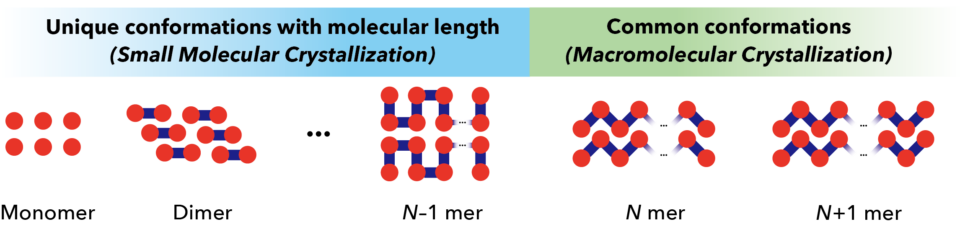
When molecules form a crystal, the way in which they arrange themselves can affect many different physical properties, including melting point, folding, and light absorption. For polymer molecules, the number of repeating subunits in the polymer chain can affect this molecular arrangement. In general, for small chain lengths, a slight change in length can lead to entirely different molecular packing arrangements and material properties. However, there is a critical chain length above which the packing arrangement stays the same for different chain lengths. Investigating this critical length is key for understanding how material properties change with the transition from small, individual molecules to large polymer macromolecules. However, it is challenging to investigate these changes due to the difficulty of synthesizing and crystallizing samples purely consisting of a discrete chain length.
In this collaborative work between ICReDD, Hokkaido University and Kanazawa University, researchers investigated the critical chain length for a polyketone compound based on an acetylacetone derivative. The benefit of using such a polyketone is that the polymer chain can be extended in a controlled, selective fashion from only one end of the chain. This enabled researchers to synthesize pure samples of molecules with a discrete chain lengths in the range of 2-20 repeating subunits. Utilizing single crystal and powder x-ray diffraction (XRD) data, the team found the critical chain length was a surprisingly short five subunits. Below this length, distinct crystal structures were determined for each chain length. At or above this length, XRD patterns were obtained indicating a helical structure. Calculations of an infinitely long chain also showed a helical structure, supporting the idea that packing structure does not change beyond the critical chain length of five. This determination aided in explaining melting point behavior. Unpredictable melting point changes occurred below the critical chain length and were attributed to the different molecular packing arrangements. As the polymer chain increased above the critical length, the melting point changes became more stable, gradually increasing with chain length.
This approach could be extended to other polymers in order to understand property changes and determine the optimal chain length of a material for a given application.