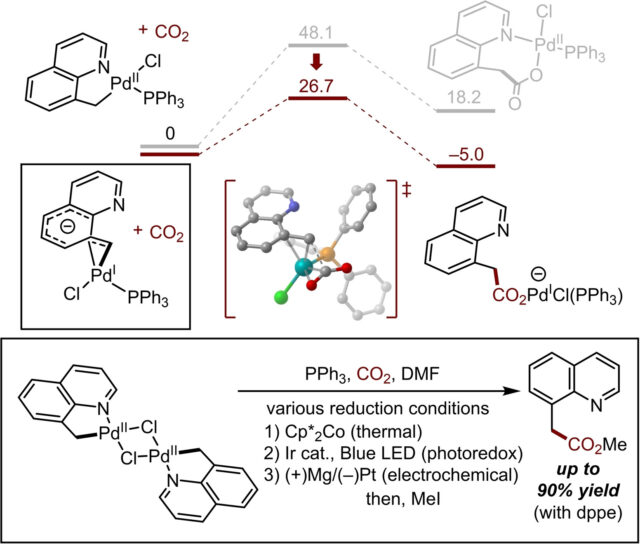
Researchers at ICReDD have reported a new reaction for carboxylation of a palladacycle that proceeds via C(sp3)–H bond activation. This synthesis was guided using insight gained from theoretical calculations using the artificial force induced reaction (AFIR) method. Reduction of the palladium complex from Pd(II) to Pd(I) was found to be theoretically crucial for the C–C bond forming step with CO2. This information enabled researchers to achieve the desired carboxylation of palladacycles in experiment under various reductive conditions, such as using Cp*2Co, photoredox catalysts, or electrochemical conditions.
By utilizing the reaction path predicted by AFIR as a guide, researchers were able to greatly accelerate the development of this transition metal complex-based reaction. This research will facilitate further development of the long sought-after catalytic C(sp3)–H bond carboxylation. More information can be found in the paper by W. Kanna, Y. Harabuchi, S. Maeda, and T. Mita et al. (DOI: 10.1002/asia.202100989).